Draw The Lewis Structures Of F2, O2 And N2. Which Statement Is True?
Tabular array of Contents
What is Electron Dot Structure?Recommended VideosHow to Draw Electron Dot Structures?How to Draw Lewis Structures?Lewis Structure ExamplesSolved Examples
What is Electron Dot Structure?
Electron dot structures or Lewis dot formula can exist fatigued if the molecular formula of the compound is known.
It defines the nature of bond and position of atoms of the molecule which are connected in the molecule.
The representation of molecules in Lewis electron dot construction or just a Lewis structure is in award of the American chemist Gilbert Newton Lewis.
Lewis dot structures likewise called electron dot structures are diagrams that describe the chemic bonding betwixt atoms in a molecule. They besides display the total number of lone pairs present in each of the atoms that plant the molecule. Lewis dot structures are unremarkably referred to as electron dot structures or Lewis structures. Lewis defined a base of operations equally an electron pair donor and an acrid as an electron pair acceptor.
Lewis Dot Structure
Lewis dot structures reverberate the electronic structures of the elements, including how the electrons are paired. Lewis structures are a useful mode to summarize certain information almost bonding and may be thought of every bit "electron bookkeeping". In Lewis dot structures each dot represents an electron. A pair of dots between chemical symbols for atoms represents a bond.
Recommended Videos

How to Draw Electron Dot Structures?
A Lewis Electron Dot Formula comprises one dot for every valence electron and an element'due south symbol. Stages to articulate the electron dot formula are stated below. Note down a skeletal structure displaying a realistic bonding blueprint by means of only the chemical element symbols.
Pick upwardly every valence electrons from every atom and toss them into a make-believe container that we can term an electron pot.
Make use of the Northward – A = South equation to get to know the number of bonds
- The sum of the number of valence electrons is equal to Due north
- The number of valence electrons in electron pot is A.
- The number of electrons shared in the molecule equals S
- Distribute the electrons from electron pot to account for the bonds
- Distribute the rest of the electrons
How to Draw Lewis Structures?
A Lewis electron dot structure describes the bonding atoms, the number of bonds in the molecule, and the lone pairs left in the bonding atoms.
The steps that must be followed while drawing a Lewis structure are listed beneath.
- Start, the total number of valence electrons present in the molecule is calculated by calculation the private valencies of each atom.
- If the molecule is an anion, extra electrons (number of electrons added = the magnitude of negative charge) are added to the Lewis dot construction.
- When cationic molecules are considered, electrons are subtracted from the full count in gild to make up for the positive charge.
- The least electronegative atom is made the central atom of the molecule or ion.
- The atoms are at present connected via single bonds.
- Now, the lone pairs of electrons are assigned to each atom belonging to the molecule. Commonly, the lone pairs are assigned to the most electronegative atoms starting time.
- Once the alone pairs are assigned, if every atom does non take an octet configuration, a double or triple bond must be drawn to satisfy the octet valency of each cantlet.
- If required, a lone pair can be converted into a bond pair in order to satisfy the octet rule for 2 atoms.
Information technology is important to note that only the valence electrons are considered while drawing Lewis dot structures and the electrons that practice not vest to the outermost shell are ignored.
Lewis Structure Examples
The Lewis electron dot structures of a few molecules are illustrated in this subsection.
1. Lewis Construction of CO2
- The central atom of this molecule is carbon.
- Oxygen contains vi valence electrons which form 2 lone pairs. Since it is bonded to only one carbon atom, it must form a double bond.
- Carbon contains iv valence electrons , resulting in goose egg lone pairs. Therefore, it is doubly bonded to each oxygen atom.
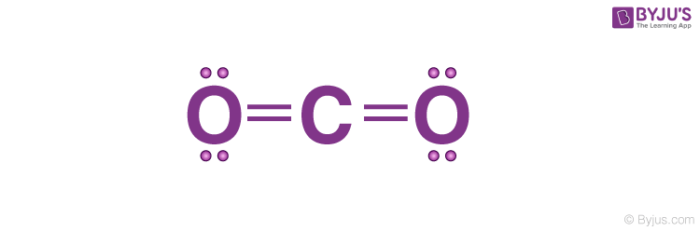
ii. Lewis Structure of O2
- An atom of oxygen contains 6 electrons in the valence trounce.
- Four of the valence electrons be in lone pairs, implying that the oxygen atom must participate in two single bonds or one double bond in order to achieve an octet configuration.
- Since in that location are only ii oxygen atoms in an O2 molecule, the atoms class a double bond resulting in the following Lewis electron dot structure.

3. Lewis Structure of CO (Carbon Monoxide)
- A carbon monoxide molecule consists of one carbon cantlet and one oxygen cantlet.
- The carbon atom requires four electrons to obtain octet configuration whereas the oxygen atom requires 2.
- Therefore, the valency is satisfied via the donation of a lone pair of electrons for bonding by the oxygen cantlet.
- The resulting Lewis electron dot structure displays a triple bond connecting a carbon and an oxygen atom, each holding a lone pair of electrons.

Solved Examples
Problem-1: In terms of electron dot formulas, define the electron structure of the carbonate ion CO 3 two- .
Solution:
One potential electron dot formula for the carbonate ion is

Since all carbon-oxygen bonds are likely to be equal, the electron structure in resonance terms is shown below.
Over the region of all three carbon-oxygen bonds, one electron pair is delocalized.
Trouble-2: Cyclopentane – What volition exist the formula and electron dot structure of this element?
Solution:
Single bonds connect five carbon atoms of cyclopentane in a cyclic form. The molecular formula of cyclopentane is C 5 H ten . The electron dot construction and structural formula of cyclopentane are articulated underneath.

Thus, the concepts related to Lewis dot structures are discussed briefly in this article along with a few examples.
Frequently Asked Questions – FAQs
What electrons do Lewis structures evidence?
The outermost central level of free energy-containing electrons is called the level of valence and includes electrons of valence. Lewis symbols are diagrams showing the number of valence electrons of a specific element with dots indicating lone pairs.
How do electron dot structures represent shared electrons?
Electron dot structure-valence electrons are represented by dots effectually the symbol of chemical science. Electrons sharing — covalent bonding — electrons sharing is the glue that holds atoms together.
What is the purpose of Lewis structures?
The aim of Lewis structures is to provide a simple fashion for chemists to view molecules that allows authentic predictions nearly the actual molecules and structure and backdrop to exist made.
What is the Lewis dot construction in chemistry?
Lewis structures, also known as electron-dot or electron-dot diagrams, are diagrams showing the bonding between a molecule's atoms and the lone pairs of electrons that may occur in the molecule.
What is the Lewis construction of ammonia?
Ammonia has the NH3 equation. It is extremely water-soluble because it is a polar material. For a molecule, the Lewis structure is the total valence electrons in the molecule. Ammonia has viii valence electrons.
What is the best Lewis structure?
The response to this question depends on how you interpret the term "best." Equally described on folio 73 of the course reading, the lewis construction with formal charges closest to 0 is "best" in the sense that information technology makes a greater contribution to the resonance mixture than the other two resonance structures.
Why are Lewis structures important?
Second, the Lewis structure'due south value. For the prediction of geometry, polarity and reactivity of (in)organic compounds, Lewis structures are actually very important. The Lewis construction is drawn for private atoms by putting a dot for each available valence electron around the atom.
To learn more about this topic and other related topics, register with BYJU'S and download the mobile application on your smartphone.
Source: https://byjus.com/chemistry/lewis-dot-structures/
Posted by: kohndeabinder.blogspot.com


0 Response to "Draw The Lewis Structures Of F2, O2 And N2. Which Statement Is True?"
Post a Comment